The Science

Overview
C-dots (carbon dots) are a novel class of carbon-based spherical nanomaterials with diameters less than 10 nm. They are present in the nature and have been found in honey and beer. With abundant laboratory techniques, c-dots have also been synthesized with diverse carbon-based precursors. The synthetic approaches of carbon dots are generally divided into top-down and bottom-up approaches mediated by hydrothermal/solvothermal, microwave, ultrasonication, laser ablation and arc discharge. In recent years, carbon dots gradually draw much attention due to their unique properties and numerous promising applications.
Properties
C-dots (carbon dots) possess the following properties:
- C-dots belong to the smallest carbon-based nanoparticle class. Considering the nanoscale, C-dots have many unique optical, catalytic and electrical properties. Carbon dots are well characterized for their excellent photoluminescence that is tunable according to the requirement for different applications.
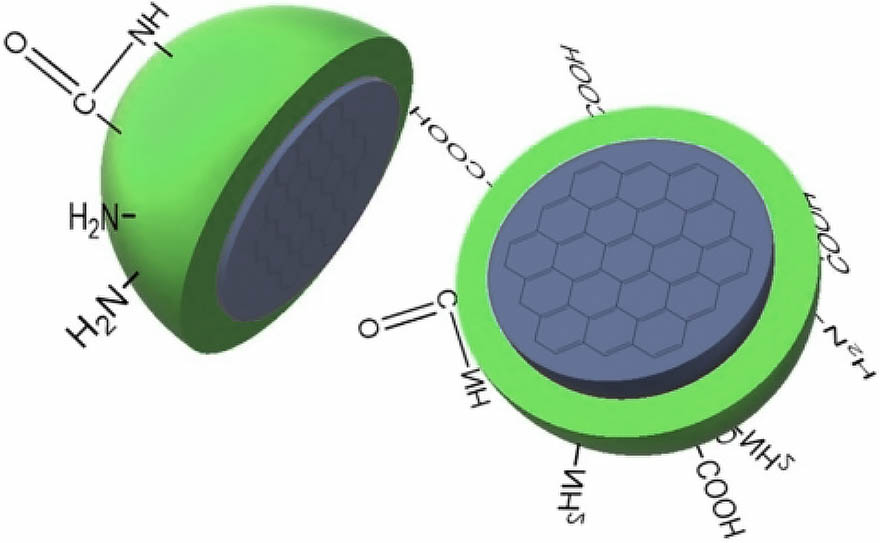
- C-dots are spherical nanoparticles. They have large surface area, high surface to volume ratio and abundant functional groups, which are beneficial for the covalent conjugation with various drug molecules.
- Due to the presence of various functional groups on the surface, the surface of C-dots is full of electron donors and acceptors, which is advantageous for catalysis.
- C-dots are biocompatible, which is supported by many in vitro and in vivo tests, as their precursors are carbon based instead of heavy, toxic metals.
- Different from traditional quantum dots, C-dots can highly disperse into water and even organic solvents with specific preparation methods.
- C-dots usually carry negative surface charge due to the rich carboxyl and or hydroxyl groups on the surface and have shown a great thermoelectric effect.